Diversity of Klebsiella Surface Antigens O and K
*Note: This hosting platform doesn't permit italicization of the genus name in this article header.
01 — Objective: To strengthen a grant proposal by creating tailored, comprehensive visual representations of the structural diversity of Klebsiella surface antigens, while indicating their localization within the bacterial cell membrane.
02 — Material:
Many Klebsiella species can be found in low numbers in a healthy human microbiome; still, opportunistic agents within the genus, including members of the Klebsiella pneumoniae species complex, Klebsiella oxytoca, and several others, represent long-standing threats to immunocompromised and otherwise vulnerable individuals. These conditional pathogens are causative agents of a range of healthcare-associated infections, including nosocomial pneumonia, catheter-associated urinary tract infections (CAUTIs), surgical site and wound abscesses, and fatal sepsis.
In recent years, emerging hyper-virulent (hvKp) and multi-drug
resistant (MDR/cKp) Klebsiella strains have also begun to
affect immunocompetent individuals, and now pose a mounting worldwide
health concern. These infectious Klebsiella species possess an
arsenal of virulence factors that contribute to pathogenicity and
assist in evasion of the host immune response.
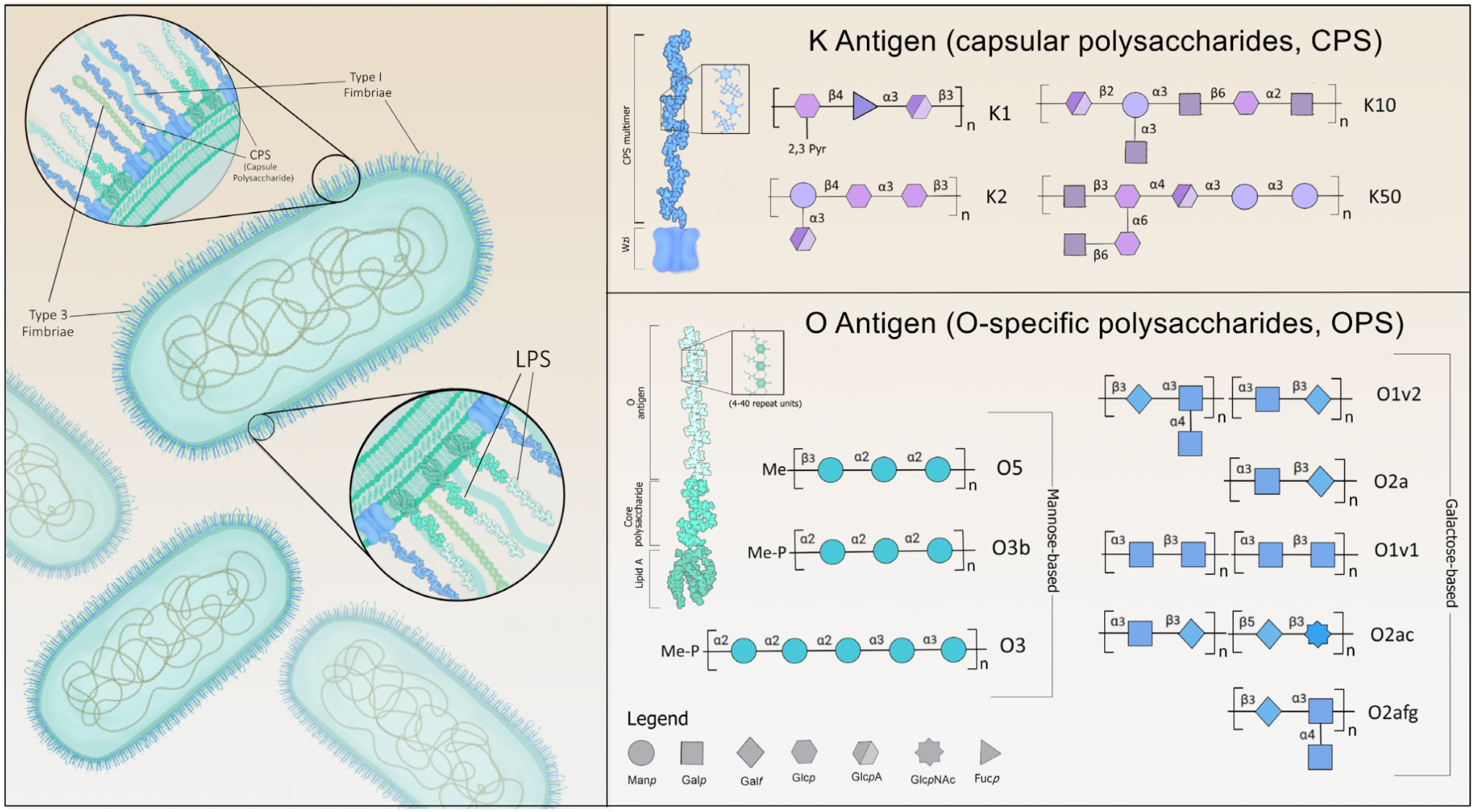
Klebsiella K
antigens (capsular polysaccharides) are a diverse group of
surface molecules containing 77-79 serologically-characterized
structural variations. Broadly, K antigens help protect the bacteria
from the host immune response at multiple levels, helping the
bacteria to avoid detection, evade cell-mediated immunity, and
block innate immune signaling cascades. The K antigen group
has a hand in reducing engulfment of the bacteria by epithelial
cells, avoiding phagocytosis by macrophages and neutrophils,
interrupting NF-kB signaling, and triggering cell death and
cytotoxicity in host cells. In hvKp strains specifically, key K
antigens, such as K1 and K2 (Figures 1 and 2), help the opportunistic
pathogen evade clearance by the host by conferring increased
resistance against two of these facets of the immune response:
complement-mediated killing and phagocytosis. Less
well-understood,
serologically-identified
variations, including K5, K20, K54, K57, and K64, are also associated
with a hypervirulent phenotype.
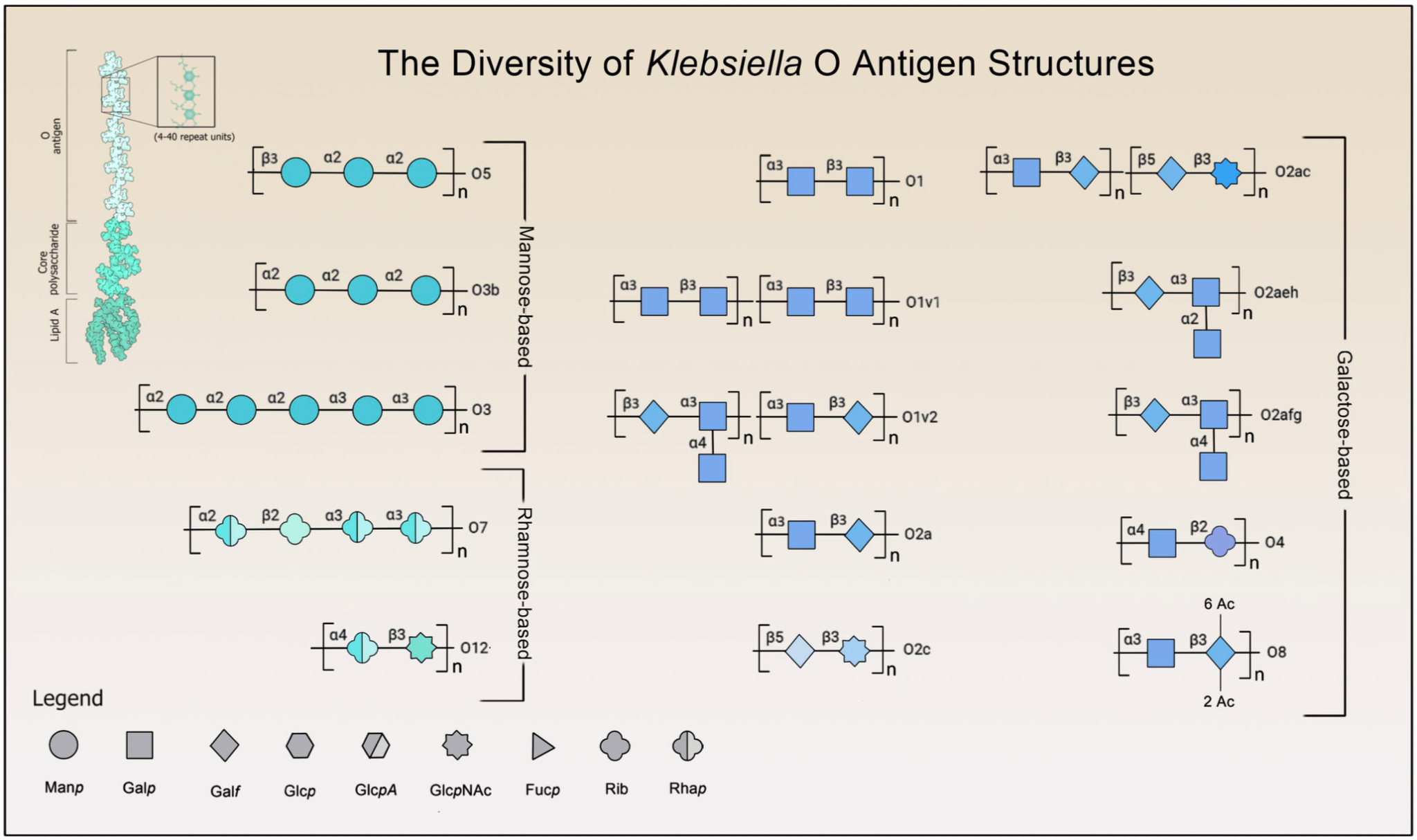
The O-specific polysaccharides (OPS), or O antigens, are a group containing around 9-13 structural variations, although O1, O2, and O3 (Figure 2) are identified in about 80% of infections. In tandem with CPS, O-specific polysaccharides help counter the host immune response by suppressing TLR-signaling and facilitating resistance to complement-mediated killing through interference with C3b deposition. The O1 variant is often seen in hvKp infections along with K1 and K2; at present, O1, O2, O3, and O5 are represented across emerging MDR/cKp strains. Altogether, the structure of these factors varies meaningfully across distinct strains, contributing in part to the rapidly developing pathologies seen in MDR, hvKp, and convergent (MDR-hvKp) species.
Over the past two decades, multi-drug resistance has been increasingly observed as a regular and concerning feature of severe human infection by opportunistic and hyper-virulent Klebsiella species. The spread of drug resistance forebodes an eventual narrowing of treatment options for Klebsiella infection, while, in parallel, an increase in hyper-virulence abets a concurrent widening of the susceptible population. The role of surface antigenic variation in individuating MDR and HV-specific pathology warrants further characterization of these rapidly-evolving virulence factors. Exploration of novel therapeutic agents and vaccines targeting these antigens denotes a promising strategy to preempt this rising global health threat.

03 — Impact: Directly supported successful acquisition of funding that initiated a new research trajectory for the client’s laboratory. The figures served as lasting reference material for ongoing project documentation and presentations.
Post a comment